Fastidious readers of my summaries of gout research studies might have noticed that whenever urate is mentioned, I almost always change it to uric acid.
This is *not* because I think that they are the same thing. It is because, for the layman, for the average gout sufferer, i.e. for 99% of visitors to GoutPal, they can be treated as the same.
I believe that gout sufferers have enough to worry about without having to learn lots of new terms. It is good to understand the inner workings of gout. Knowledge is power, and if you understand how gout works, you are better placed to control it. However, as a medical problem, we are up against the intricacies of biochemistry and human biology. Some of these processes are very complicated, with every other word an unfamiliar medical or biological term.
Before I continue, I should explain that I have written this article in response to an off-topic comment on another thread. I will move that comment here as soon as I publish this. This will make the discussion a little disjointed, but it should still be readable. If you prefer, go and read the first comment now, then return here to read my explanation of urate vs uric acid.
Urate Explanation
I have to be honest and admit that much of it is beyond me. Even if I can grasp some of the principles, it is quite another thing to be expert enough to be able to explain it in easily understandable terms. If I thought it would help manage gout, then I might make more effort. However the simple truth is, if you control your uric acid by reference to your blood test results, you do not need to understand the different forms that uric acid can take. Just set your limit at 5 mg/dL (0.30 mmol/l), test at least once a year, and take responsibility for your uric acid level, and you will control gout.
If you must understand the differences between the states of uric acid, including combination with other substances as urate, I will try to give pointers to the relevant science. In turn, you can check out the science and explain the processes in layman’s terms once you are expert enough.
If enough people express an interest in understanding the principles behind urate vs uric acid, then I will prepare a clearer, illustrated article for the knowledgebase on the main gout website.
What Is Urate?
Urate is uric acid combined with another substance, usually sodium. Most commonly it forms MonoSodium Urate, which is usually referred to as MSU. Other compounds are formed occasionally, as uric acid bonds with other elements. There is biochemistry involved here, and it gets complicated. The point to understand is that it is not the same as inorganic chemistry.
Early high school science includes chemical reactions where acids bond with bases. These tend to create very strong bonds that require enormous energy, or other reagents to break the bond and reduce the component elements.
Organic chemistry is much more fluid. The hydrogen ion of uric acid will attract unbonded sodium and form sodium urate in different forms.The most common of these is monosodium urate, where the mono means one hydrogen ion is linked. Researching this process is made more difficult by the fact that there are different notation methods for organic chemistry. A common expression describes the process as:
NaC5H3N4O3(s) <—–>(equilibrium) Na+(aq) + C5H3N4O3-(aq)
The aq refers to aqueous solution – i.e. uric acid dissolved in our blood or joint fluid. Thus, we see that monosodium urate is in equilibrium with dissolved uric acid and dissolved sodium ions. Equilibrium means that uric acid will move between different states dependent mainly on concentration, but also affected by factors such as temperature.
If you search for the equation, you will find lots of K12 chemistry classwork. Apparently, the uric acid to urate exchange is a popular high school problem. You can even find some kid’s homework with an explanation of the molecular weights, or as they more correctly put it “molar masses”
I’m sure this is fascinating to a chemist, but I thought molar masses were what the tooth fairy paid for.
Anyway, the important guiding notion comes in the form of Le Chatelier’s Principle. Your homework is to present me with an explanation that a six year old could understand (with bonus points for the six year old child reference).
All that gout patients need to understand is their uric acid number. At 5 or below, the uric acid stays happily dissolved in your blood. That extra hydrogen ion is probably useful in equilibrium, but if I suggest it might bond with an appropriately charged oxygen ion for the benefit of our health, I’m bound to get radical abuse (NY GoutPal in-joke).
Le Chatelier guides us to understand that, as our uric acid number moves up the scale towards 7, more uric acid will link with free sodium (most commonly, as sodium is usually common in our bodies. Other ions are available). The resulting MSU is less soluble than pure uric acid, but even that will crystallize if the concentration gets high enough.
In a lab, if we lower uric acid concentration, Le Chatelier comes to our rescue, and to maintain the equilibrium, the bond breaks, and uric acid floats it’s own way, with sodium ions floating their own way.
In our bodies, there are many more processes occurring, but the basic principle holds – lower the amount of uric acid dissolved in your blood, and MSU will change back to uric acid and sodium ions. If your kidneys are working OK, the excess uric acid should be excreted.
This process is why allopurinol works. It does not directly dissolve uric acid crystals. It creates a lower uric acid environment in which MSU de-combines. In the separated state, the solid crystal returns to the dissolved fluid. Gout symptoms go away.
Unfortunately, our bodies are something of a battleground, so other processes make a mess of our simple uric acid/MSU model.
Once solid crystals form, they get encased in white blood cells. This is the fundamental process of gout. White blood cells attack the crystals, and engulf them. They call in reinforcements and that process (phagocytosis) produces the incredible gout pain with which we are familiar. In the engulfed state, the crystals are invisible to the immune system, so the gout flare diminishes. When this first happens, it is quite easy to reverse. The dead cells are semi-permeable, so in a low uric acid environment, the sodium ion will break free from the MSU crystal, and pure uric acid will dissolve away. At the start of this process, the coating of white blood cell will shed, exposing the partially dissolved crystal. If uric acid concentration is low enough, the sodium ions will stay away, and the crystal will dissolve completely. If uric acid concentration is borderline, then the Le Chatelier’s equilibrium principle will force new MSU bonds, with new crystals. At around 7mg/dL, you create an environment of constantly dissolving and reforming crystals. That is why, if your doctor under-doses your allopurinol, he is a bastard.
I accept that this is slightly more than uric acid moving from pure liquid form to pure crystals form. To the layman, does it really matter that sodium ions are involved? It is uric acid crystals, even if you want to call it urate when it is solid.
The above represents my unexpurgated explanation of the urate vs uric acid process. It is far from perfect. Let us treat it as a starting point. If there is sufficient interest, tell me which points need clarifying further, and I will build a proper easy-to-understand reference resource.
Long Term Urate Factors
Before I finish, I’d like to touch on the problems of gout that has been left uncontrolled for a long time.
The equilibrium processes I have described apply to free-floating uric acid, and crystals that are free-floating or recently encased by white blood cells. Over time, the encased urate crystals build to form less penetrable masses. In addition to collections of encased crystals that form tophi under the skin, we have severe skeletal problems.
As I reported recently, the white blood cells that should be destined to repair and grow tendons, cartilage, and bone, get caught up in the gout battle. Any uric acid crystals near the skeleton are likely to get encased within bones, tendons and ligaments. In some cases, the joint crumbles, in other cases we get masses of crystals bonding with dead cells as outgrowths on the skeleton. Similarly with unattached tophi under the skin. These lumps grow bigger, and so crystals become more isolated.
I have this experience personally, with a tophi near my right elbow that has shrunk since I started allopurinol, but is still fairly prominent. There is certainly a question mark over the total solubility of old encased crystals. The process of dissolving uric acid crystals is very dependent on how low we can get uric acid concentration. I always recommend searching for lower faster, as this reveals my favorite gout research. It proves that tophi can be dissolved, and it guides us to go for the lowest concentration possible. This is reinforced in recent reports from pegloticase (Krystexxa) usage. With that gout medication, uric acid is reduced to almost zero, and the speed with which visible tophi dissolve is amazing.
It might be that we can do nothing for invisible tophi – the uric acid crystals that have fused with the skeleton over the years. I have to remain philosophical about that. If they cannot be dissolved, then at least they are unlikely to be active gout triggers. I may have to live with some old, embedded crystals. I may have to live with some joint damage. I must use that to drive my education process. For newly diagnosed gout patients, I say to you, please do not worry if it is called uric acid or urate – just get it safe before your skeleton suffers.
“Those undissolved uric acid crystals will still be hanging around to cause more gout pain in future.”
Sorry, Keith, for throwing in another monkey wrench.
Neither YOU, nor the MEDICAL INDUSTRY, want to neither look at nor deal with the facts: URIC ACID (UA), (a solid substance) is an ACID which is in the blood and is being transferred through our system via the blood. The undissolved crystals outside the blood, (what are called: TOPHI) are MONO-SODIUM URATE (MSU), the salt of uric acid. They are neither chemically nor physically the same.(If you need me to prove that, I can supply you the facts about those two chemicals.) Anyone who treats them the same, is saying that cooking salt (NaCl) is the same as hydrochloric acid (HCl) While we shove salt (NaCl or KCl=Potassium Chloride) down our throat daily on a regular basis, I doubt very much that you would drink HCl for very long.
(Funny thing though, we have it in our stomachs to regulate our digestion. Have you ever heard of “Heartburn”? What a crazy word for what it is. That’s when too much acid in your stomach comes up that pipe and seems to burn down your throat, not your heart.)
To get back to UA and MSU: UA production in the blood is being controlled/reduced by using Allopurinol or Uloric. Probenecid takes the existing UA out of our body via the kidneys; it does not reduce the production of the acid. Hence, it makes more sense to either use Allo or Uloric to treat the cause, not the symptom, the preferred method of the MEDICAL INDUSTRY and the PHARMACEUTICAL INDUSTY.
I challenge the statement that Allo or Uloric dissolves MSU crystals. In order to dissolve something you must have a solvent. So what is that solvent which dissolves MSU? I have yet see a concise paper, experiment, explanation of that process. Nobody is going to convince me that those MSU barnacles, attached to the bones/joints will readily let go and migrate back into the blood as AU. I can still hear the scratching and see my doctor’s knife when she scraped those tophi of my index finger.
Keith, I neither wish to criticize nor ridicule your invaluable work you have performed over the last 6 years for the Gouty Community (and specially for me. Thanks to your guidance, admonitions, and hammering into our brains the importance of the low UA level of 5mg/dL, I have been without a flare or gout attack for more than 20 months now, the longest period in my 28 year gout history.) It just has bugged me to no end that the gurus of the MEDICAL and PHARMACEUTICAL INDUSTY have never clarified the distinction and interdependence of UA and MSU.
Hans, I hope you do not mind me moving your comment like this, but the original location was not good enough for this topic. Let’s forget the problems of moderating discussions, and move to the heart of the problem.
It is my belief that the crux of the matter is the difference between organic and inorganic chemistry. In the latter, I have distant memories of covalent bonds and the like whereby reactions take place that are rarely, or not easily, reversible.
I believe the organic reactions involved with MSU and uric acid are completely different. Guided by laws of equilibrium, we have a state where, when one factor gets adjusted, other reactions occur to compensate. Thus, if we set the concentration of uric acid and sodium, when temperature moves down, more MSU is formed. Raise the temperature, and MSU dissociates back to dissolved uric acid and dissolved sodium.
If I have got this wrong, I would prefer a proper explanation why that is so.
I accept that it is wrong to say that allopurinol will dissolve uric acid crystals. It is true that allopurinol, or anything else that lowers uric acid, or changes the equilibrium, creates the environment where MSU or uric acid crystals must dissolve.
I believe this is why so many gout studies simply use the term urate, or urate pool. They are considering the entire system of uric acid in solution, in floating crystals, and in fixed deposits. By managing that pool, we manage gout.
As for the barnacles, I hope these form a very small part of that pool. At least small enough to be insignificant when blood uric acid is maintained at a safe 5mg/dL long term. I look forward to advances in technology being able to measure that. Even better, that advance may make it possible for much earlier detection of deposits near the skeleton. In future, we may be able to reverse the MSU process before it has damaged the skeleton.
It’s gone rather quiet in this discussion. Is the science numbing a few brains?
To liven it up, I spotted a relevant photograph today, together with an explanation that included: “Even the largest tophi may be dissolved with use of appropriate medication and with sufficient patient adherence.”
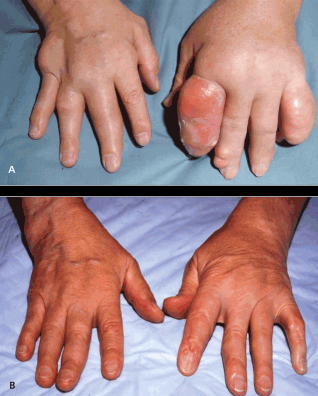 More information when you click the photograph.
More information when you click the photograph.